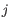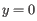The GLM model
Fitting a GLM to expression data
It is easiest to understand how the GLM model works through an example. Imagine an experiment looking at the effect of two drug treatments while controlling for gender:
- Test differential expression due to Treatment with three groups: drugA, drugB, placebo
- While controlling for Gender with groups: Male, Female
In an abuse of mathematical notation, the underlying GLM for each gene looks like
where ![]() is the expression level for the gene in sample
is the expression level for the gene in sample ![]() ; the combined term
; the combined term
![]() describes an arbitrarily chosen baseline expression level (of males being given a placebo); and the other terms
describes an arbitrarily chosen baseline expression level (of males being given a placebo); and the other terms
![]() ,
,
![]() and
and
![]() are numbers describing the effect of each group with respect to this baseline. The
are numbers describing the effect of each group with respect to this baseline. The
![]() accounts for differences in the library size between samples. For example, if a subject is male and given a placebo we predict the expression level to be
accounts for differences in the library size between samples. For example, if a subject is male and given a placebo we predict the expression level to be
If instead he had been given drug B, we would predict the expression level ![]() to be augmented with the
to be augmented with the
![]() coefficient, resulting in
coefficient, resulting in
We assume that the expression levels ![]() follow a Negative Binomial distribution. This distribution has a free parameter, the dispersion. The greater the dispersion, the greater the variation in expression levels for a gene.
follow a Negative Binomial distribution. This distribution has a free parameter, the dispersion. The greater the dispersion, the greater the variation in expression levels for a gene.
The most likely values of the dispersion and coefficients,
![]() ,
,
![]() and
and
![]() , are determined simultaneously by fitting the GLM to the data. To see why this simultaneous fitting is necessary, imagine an experiment where we observe counts {15,10,4} for Males and {30,20,8} for Females. The most natural fit is for the coefficient
, are determined simultaneously by fitting the GLM to the data. To see why this simultaneous fitting is necessary, imagine an experiment where we observe counts {15,10,4} for Males and {30,20,8} for Females. The most natural fit is for the coefficient
![]() to have a two-fold change and for the dispersion to be small, but an alternative fit has no fold change and a larger dispersion. Under this second fit the variation in the counts is greater, and it is just by chance that all three Female values are larger than all three Male values.
to have a two-fold change and for the dispersion to be small, but an alternative fit has no fold change and a larger dispersion. Under this second fit the variation in the counts is greater, and it is just by chance that all three Female values are larger than all three Male values.
Refining the estimate of dispersion
Much research has gone into refining the dispersion estimates of GLM fits. One important observation is that the GLM dispersion for a gene is often too low, because it is a sample dispersion rather than a population dispersion. We correct for this using the Cox-Reid adjusted likelihood, as in the multi-factorial edgeR method [Robinson et al., 2010]. To understand the purpose of the correction, it may help to consider the analogous situation of calculation of the variance of normally distributed measurements. One approach would be to calculate
![]() , but this is the sample variance and often too low. A commonly used correction for the population variance is:
, but this is the sample variance and often too low. A commonly used correction for the population variance is:
![]() .
.
A second observation that can be used to improve the dispersion estimate, is that genes with the same average expression often have similar dispersions. To make use of this observation, we follow [Robinson et al., 2010] in estimating gene-wise dispersions from a linear combination of the likelihood for the gene of interest and neighboring genes with similar average expression levels. The weighting in this combination depends on the number of samples in an experiment, such that the neighbors have most weight when there are no replicates, and little effect when the number of replicates is high.
When estimating dispersion, we use the following strategy:
- We sort the genes from lowest to highest average logCPM (CPM is defined also by the edgeR authors).
For each gene, we calculate its log-likelihood at a grid of known dispersions. The known dispersions are
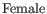 , where
, where
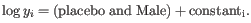 such that there are 11 values of i in total. You can imagine the results of this as being stored in a table with one column for each gene and one row for each dispersion, with neighboring columns having similar average logCPM (because of the sorting in the previous step).
such that there are 11 values of i in total. You can imagine the results of this as being stored in a table with one column for each gene and one row for each dispersion, with neighboring columns having similar average logCPM (because of the sorting in the previous step).
- We now calculate a weighted log-likelihood for each gene at these same known dispersions. This is the original log-likelihood for that gene at that dispersion plus a "weight factor" multiplied by the average log-likelihood for genes in a window of similar average logCPM. The window includes the 1.5% of genes to the left and to the right of the gene we are looking at. For example, if we had 3000 genes, and were calculating values for gene 500, then
 , so we would average the values for genes 455 to 545. The "weight factor" = 20 / (numberOfSamples - number of parameters in the factor being tested). This means that the greater the number of samples, the lower the weight, and the fewer free parameters to fit, the lower the weight.
, so we would average the values for genes 455 to 545. The "weight factor" = 20 / (numberOfSamples - number of parameters in the factor being tested). This means that the greater the number of samples, the lower the weight, and the fewer free parameters to fit, the lower the weight.
- We fit an FMM spline for each gene to its 11 weighted log-likelihoods. Our implementation of the FMM spline is translated from the Fortran code found here https://www.netlib.org/fmm/spline.f. This spline allows us to estimate the weighted log-likelihood at any dispersion within the grid i.e., from
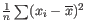 to
to 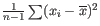
- We find the dispersion on the spline that maximizes the log-likelihood. This is the dispersion we use for that gene.
Downweighting outliers
It is often difficult to detect outliers. Therefore, instead of removing putative outliers completely from the GLM fit, we give them "observation weights" that are smaller than 1. These weights reduce the effect of the outliers on the GLM fit.
Outliers can have two undesirable effects. First, genes with outliers are more likely to be called as differentially expressed because a sample with anomalously high expression is hard to reconcile with no fold change. Second, outliers can prevent differentially expressed genes from being detected. This is because, when a gene has an anomalously high expression, the dispersion for that gene will typically be overestimated. Consequently the dispersion of genes with similar expression will be overestimated, such that they are less likely to be called as differentially expressed.
When downweighting outliers, the above dispersion estimation procedure is repeated 5 more times, and per-gene weights are estimated for the samples at each iteration. Note that these weights are not related to the weights in the weighted log-likelihood of the dispersion estimation procedure.
The following iterative procedure is performed to estimate the observation weights. For each gene, one iteration proceeds as follows:
- The Cook's distance is calculated for each sample. The Cook's distance for sample
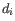 is
where
is
where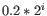
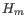 is the number of parameters in the fit.
is the number of parameters in the fit.
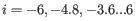 is the Pearson residual for sample
is the Pearson residual for sample 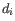 , a measure of how well the fitted model predicts the expression of sample
, a measure of how well the fitted model predicts the expression of sample 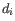 . The Pearson residual is calculated as the difference between the measured expression
. The Pearson residual is calculated as the difference between the measured expression 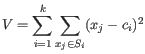 and the modeled expression
and the modeled expression  , divided by the standard deviation
, divided by the standard deviation  of the negative binomial distribution with dispersion
of the negative binomial distribution with dispersion  that has been fitted to the gene:
that has been fitted to the gene:
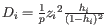
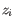 is the leverage of sample
is the leverage of sample 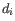 on the fit, a measure of how much sample
on the fit, a measure of how much sample 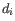 affects the fitted parameters. The leverage is the
affects the fitted parameters. The leverage is the 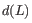 entry on the diagonal of the projection matrix
entry on the diagonal of the projection matrix 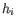 which relates measured expressions to modeled expressions:
which relates measured expressions to modeled expressions:
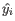 .
.
Outliers are expected to have large Cook's distances because they tend to be poorly fitted, resulting in large
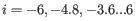 , and they are likely to disproportionately influence the fit compared to other points, leading to large
, and they are likely to disproportionately influence the fit compared to other points, leading to large 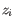 .
.
- Cook's distances are assumed to follow an F distribution
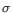 , where
, where 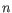 is the number of samples. For each sample, this distribution is used to calculate the probability
is the number of samples. For each sample, this distribution is used to calculate the probability 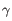 of obtaining a Cook's distance as extreme as or more extreme than the observed
of obtaining a Cook's distance as extreme as or more extreme than the observed 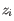 . A first approximation for the sample observation weight is
. A first approximation for the sample observation weight is 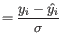
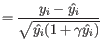
The probability cut-off of
 is set empirically from experiments on real data.
is set empirically from experiments on real data.
- This approximation is not effective in practice when outliers are present because a single outlier in a condition can influence the fit for all other samples within that condition, causing them to also exhibit large Cook's distances and appear as outliers. As a result, this approximation leads to reduced observation weights of many non-outlier samples, especially during the initial iteration of the procedure. We address this in two parts:
- We limit how much a group of samples is downweighted. For the example at the start of this section, the groups are "drug A female", "drug A male", "drug B female" etc. The allowed downweighting for a group
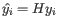 containing
containing 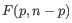 samples is:
samples is:
We hence disregard at most one sample or up to 10% of the samples from the group, whichever is larger. However, at least one sample from the group must be kept. In practice, the weights are spread across all samples: while some samples may be more heavily downweighted than others, it is uncommon for any sample to be entirely ignored, i.e., receive an observation weight
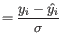 of 0.
of 0.
- We calculate a normalized skewed weight
to penalize the most extreme outliers, where
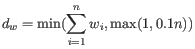
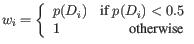 is the smallest
is the smallest 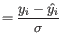 across all samples, regardless of their group.
across all samples, regardless of their group.
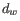 is the normalization factor such that
is the normalization factor such that
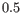 .
.
If the true outlier has a
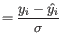 that is 10 times smaller than another sample in the same group, then
that is 10 times smaller than another sample in the same group, then 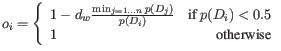 for the true outlier will also be 10 times smaller than the one for the other sample.
for the true outlier will also be 10 times smaller than the one for the other sample.
- We limit how much a group of samples is downweighted. For the example at the start of this section, the groups are "drug A female", "drug A male", "drug B female" etc. The allowed downweighting for a group
- The final observation weights are:
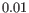
where
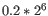 is a normalization factor used to ensure that at most one sample, or 10% of the total number of samples
is a normalization factor used to ensure that at most one sample, or 10% of the total number of samples 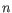 are disregarded from the entire dataset:
are disregarded from the entire dataset:
- The GLM is re-fit, using these weights.
The above steps are repeated until no observation weight changes by more than ![]() , or a weight that was
, or a weight that was ![]() becomes 1 in the following round, or a weight that decreased by more than
becomes 1 in the following round, or a weight that decreased by more than ![]() in the previous round increases by more than
in the previous round increases by more than ![]() in the next round. These stopping criteria are needed because the weights do not converge: when a weight becomes sufficiently small, the leverage component of the Cook's distance tends to zero and so the Cook's distance also becomes zero and the weight in the next iteration will be 1.
in the next round. These stopping criteria are needed because the weights do not converge: when a weight becomes sufficiently small, the leverage component of the Cook's distance tends to zero and so the Cook's distance also becomes zero and the weight in the next iteration will be 1.
Once the observation weights have been determined, the re-fitted GLM has a different likelihood than before. This is partly because the weighting is applied directly to the likelihood, and partly because the weighting affects the fitted coefficients.
The use of observation weights in the GLM is based on [Zhou et al., 2014]. The use of Cook's distance is motivated by DESeq2, which ignores genes with extreme Cook's distances when the number of replicates is small, rather than downweighting them. The form of the weights is novel to CLC Genomics Workbench.
Statistical testing
The final GLM fit and dispersion estimate allows us to calculate the total likelihood of the model given the data, and the uncertainty on each fitted coefficient. The two statistical tests each make use of one of these values.
- Wald test
- Tests whether a given coefficient is non-zero. This test is used in the All group pairs and Against control group comparisons. For example, to test whether there is a difference between subjects treated with a placebo, and those treated with drugB, we would use the Wald test to determine if the
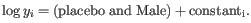 coefficient is non-zero.
coefficient is non-zero.
- Likelihood Ratio test
- Fits two GLMs, one with the given coefficients and one without. The more important the coefficients are, the greater the ratio of the likelihoods of the two models. The likelihood of both models is computed using the dispersion estimate and observation weights of the larger model. This test is used in the Across groups (ANOVA-like) comparison.
If we wanted to test whether either drug had an effect, we would compare the likelihoods of the GLM described with equation
with those in the reduced GLM with equation