Create MLST Scheme
The Create MLST Scheme tool can be used to create a scheme from scratch.
To run the Create MLST Scheme tool, go to:
Tools | Microbial Genomics Module (![]() ) | Databases (
) | Databases (![]() ) | MLST Typing (
) | MLST Typing (![]() ) | Create MLST Scheme (
) | Create MLST Scheme (![]() )
)
As input, the tool requires a set of complete isolate genomes in the form of one or more sequence lists or sequences. At least one of these genomes must be annotated with coding region (CDS) annotations. If these are not available, the Find Prokaryotic Genes tool (see Find Prokaryotic Genes) or Annotate with DIAMOND (see Annotate with DIAMOND) can be used to predict and annotate the coding regions.
In the first wizard step shown in figure 14.1 the grouping of sequences into genomic units can be controlled. This is necessary when working with genomes that span several chromosomes or several contigs for the tool to consider these as one unit. The grouping can be controlled by the Assembly grouping field:
- Each sequence is one assembly: Each individual sequence is considered a complete assembly of a genome.
- Each input element is one assembly: Each input element, i.e. input sequence or input sequence list, is considered a complete assembly of a genome.
- Group sequences by annotation type: Use annotations to group the assemblies and specify the annotation field with Assembly annotation type. Some tools, such as the Download Custom Microbial Reference Database, will automatically assign an Assembly ID that can be used for grouping. For a manual assignment of Assembly ID annotations, please see Using the Assembly ID annotation.
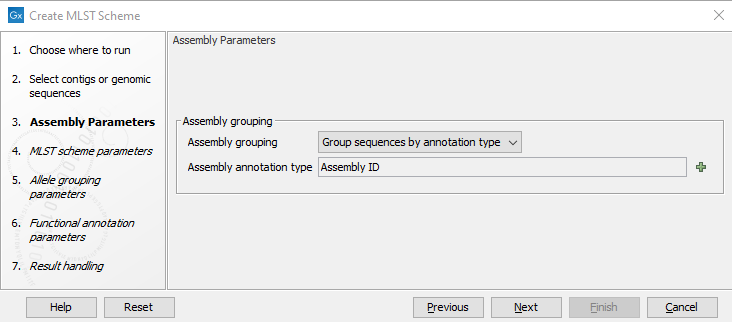
Figure 14.1: Grouping the input into assemblies.
After specifying the input, the second step is to set up the basic MLST Scheme creation parameters (figure 14.2).
The Create MLST Scheme tool works by extracting all annotated coding sequences (CDS) and clustering them into similar gene classes (loci). It is possible to specify whether we are interested in the genes that are present in some genomes (Whole genome - must be present in at least 10% of all genomes), most genomes (Core genome - must be present in at least 90% of the genomes), or a user-specified Minimum fraction.
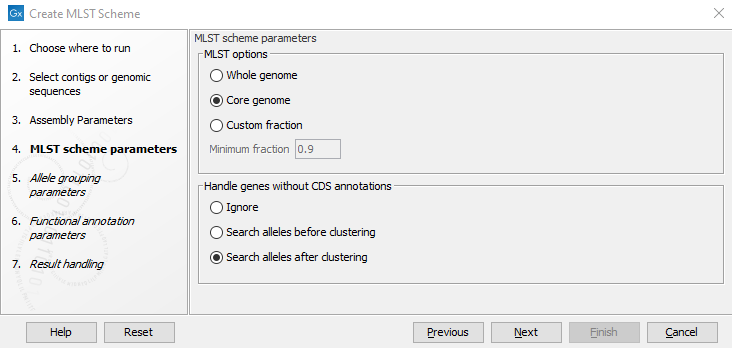
Figure 14.2: Basic options for creating an MLST scheme.
The best results are obtained by supplying genomes with proper CDS annotations. The Handle genes without annotations option controls how genomes without CDS annotations and how existing CDS may be overridden if a longer CDS from another genome exactly matches the genomic sequence.
- Ignore: Only use the existing CDS annotations as a basis for the MLST scheme construction.
- Search alleles before clustering: All of the input genomes are blasted (using DIAMOND) against the set of annotated genes, and any new genes will be added as alleles. This is a very slow, but thorough check.
- Search alleles after clustering: After clustering the genes, all of the input genomes are blasted (using DIAMOND), but only against the longest protein in each cluster.
The Allele grouping parameters step (figure 14.3) specifies how the different genes (CDS annotations) are compared to each other. DIAMOND is used for this clustering. The following can be specified:
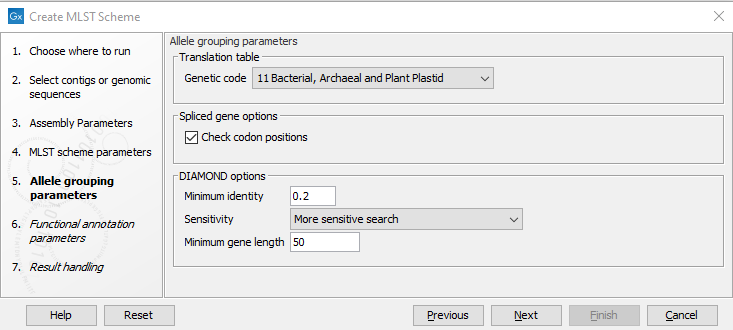
Figure 14.3: The allele grouping (clustering) options.
- Genetic code: Specify the genomic code to use for the input samples if Check codon positions is enabled.
- Check codon positions: If this is enabled, coding sequences not starting with a start codon, not ending with a stop codon or containing internal stop codons will be discarded. This can be disabled, for example to allow the construction of MLST schemes with spliced genes where each exon is considered an allele.
- Minimum identity: Set the minimum sequence identity before grouping protein sequences.
- Sensitivity: Select DIAMOND sensitivity:
- Faster search: The fastest search
- Fast search: Designed for finding hits of >90% identity
- Standard search: Designed for finding hits of >60% identity
- Mid-sensitive search: More sensitive than standard search and faster than sensitive search.
- Sensitive search: Designed for finding hits of >40% identity
- More sensitive search: Designed for finding hits of >40% identity with some motif masking disabled
- Very sensitive search: Designed for finding hits of 40% identity
- Most sensitive search: The most sensitive search
- Minimum gene length: Set this threshold to remove short genes from the resulting MLST scheme.
It is possible to decorate the alleles with information about virulence or resistance. The information can be extracted from either a ShortBRED Marker database or a Nucleotide database. These databases can be accessed using Download Resistance Database and can be provided as input to Create MLST Scheme at this step (figure 14.4).
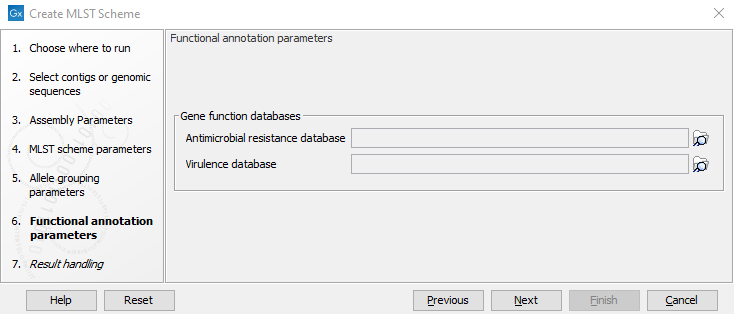
Figure 14.4: The functional annotation parameters.
